Background
Smoldering Multiple Myeloma (SMM) is an intermediate precursor phase in Multiple Myeloma (MM) evolution. SMM patients are monitored for progression to active disease, supposing such surveillance will identify myeloma-defining events (MDEs) early and consequently hasten treatment, prevent complications and improve outcomes. To what extent this approach is successful is not well established. We aimed to compare disease characteristics, end organ involvement and outcomes between SMM patients who progressed to active MM vs newly diagnosed MM (NDMM) patients.
Methods
We performed a two-center real-world case-control retrospective cohort study. Consecutive patients receiving first line treatment for MM after progressing from SMM were identified; each was matched by age and year of diagnosis (±2 years) to a patient first presenting as NDMM. SMM, MM, MDEs and response were defined by the International Myeloma Working Group (IMWG) criteria (2014, 2016); progression-free survival (PFS) and overall survival (OS) were calculated from active MM diagnosis. Data was extracted in 07/2023.
Results
We identified two cohorts of newly diagnosed MM patients, presented between 09/2008and 02/2023: Post SMM (P-SMM, N=57) and De novo MM patients (D-MM, N=57). Median follow up time was 38.6 months (IQR 23.3 - 72.7).
There were no differences in baseline demographics between gender (p=.19), and median age: 73.4 (range 42 - 88) in the P-SMM group and 72.9 (range 42 - 92) in the D-MM (p=.75).
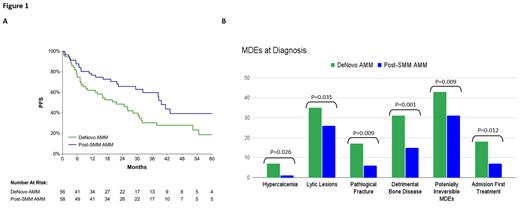
Comparing MDEs using SLiM-CRAB criteria between groups, more patients in the D-MM group presented with hypercalcemia (p=.02) and lytic lesions (p=.03). Other domains were similar between groups: anemia (p=.68), renal failure (p=.59), FLC ≥100 (p=.76), % bone marrow plasma cells (BMPC) ≥ 60 (p=.11). D-MM patients had higher ISS/R-ISS at presentation: ISS ≥ II (30 vs 11, p=.004) and R-ISS ≥ II (31 vs 13, p=.002).
Additional disease characteristics were balanced at diagnosis of active MM: heavy chain (p=.53), light chain type (p=.69), M-Protein level (p=.23), extra medullary disease (p=.14), Al amyloidosis (p=.13), and high-risk cytogenetics (p=.65).
Bone involvement was more frequent in D-MM vs P-SMM patients, with significantly more pathological fractures (p=.006), bone pain (p=.001), and a detrimental bone disease (p=.001) (defined as combination of pathological fracture and/or ≥ 3 lytic lesions) (Figure 1B).
Significantly higher rates of hospital admissions for upfront therapy initiation were seen in the D-MM group compared with P-SMM, 18 vs 7 (p=.008), as well as more potentially irreversible MDEs (pathological fractures and/or renal injury): 43 (p=.009) vs 31 (41.9%) respectively.
Treatment patterns were balanced excluding bortezomib used more often in the D-MM (p=.009) and daratumumab in the P-SMM group (p=.03).
Best response rates to upfront therapy were comparable between groups, (p=.71) as well as ≥ very good partial response (VGPR) rates (p=.88), 52.4% vs 47.6% in P-SMM and D-MM respectively.
Frequency of skeletal imaging tests up-to 2-year period prior to active MM diagnosis were associated with lower rates of detrimental bone disease (p=.006).
Despite the similar ≥ VGPR rates between cohorts, PFS was longer in the P-SMM group vs D-MM group, 39.6 vs 21.6 months, respectively (log-rank 0.01). A superior PFS in P-SMM group vs D-MM remained after correction for % BMPC at diagnosis (46.2% vs 58.8%, p=.03). OS was also longer, not reached vs 93.7 months (log-rank=.047) (Figure 1A).
Conclusions
Patients who develop active MM post SMM clinic surveillance presented with a lower disease burden, as well as reduced rates of detrimental bone disease and potentially irreversible MDEs. P-SMM patients had favorable PFS and OS rates. This may reflect effectiveness of SMM monitoring resulting in lower disease burden at time of treatment onset; favorable long-term outcomes may also be affected by variability in treatment patterns between groups. Another possible explanation is that patients with more indolent MM biology may have extended periods of precursor condition, hence increasing their likelihood to be diagnosed by incidental laboratory tests during their SMM phase.
Disclosures
Cohen:Sanofi: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; GSK: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Raanani:Janssen: Consultancy, Research Funding; BMS: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Novartis: Consultancy, Research Funding. Avivi Mazza:AbbVie: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal